
MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = n / V = mol / L Calculate. - ppt download
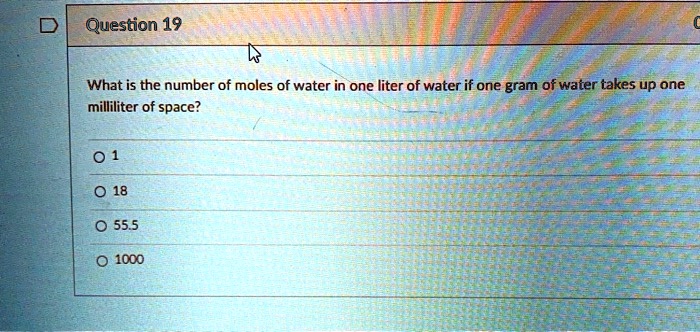
SOLVED: Question 19 What is the number of moles of water in one liter of water if one gram of water takes up one milliliter of space? 0 1 0 18 0 555 I000

SOLVED: Mole-Volume Conversions Determine the volume in liters occupied by 0.030 moles of a gas at STP. How many moles of argon atoms are present in 11.2 L of argon gas at












