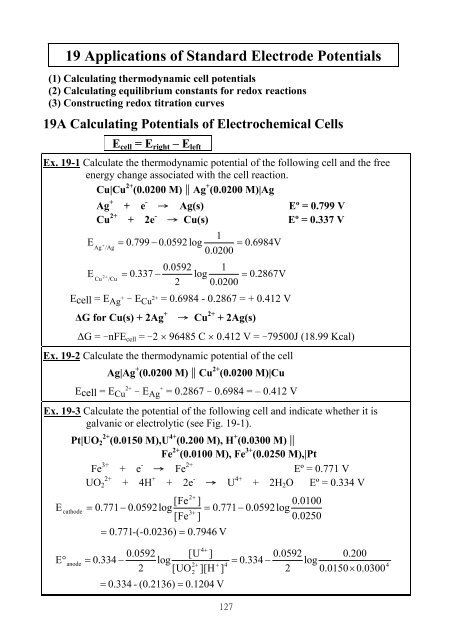
Calculate the standard electrode potential of the Ni^(2+)//Ni electrode , if the cell potential ... - YouTube
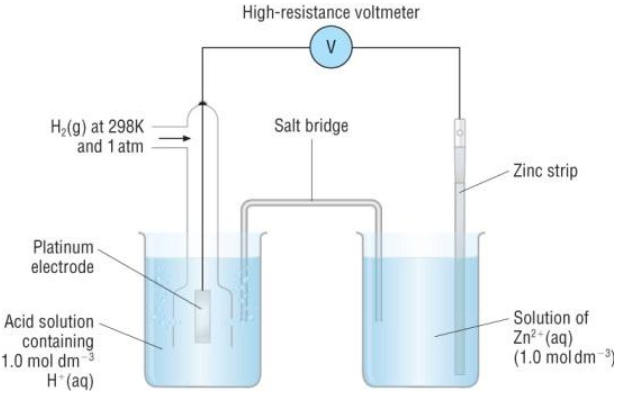
Electrode Potentials and Electrochemical Cells - Electrode Potentials (A-Level Chemistry) - Study Mind

The standard electrode potential for Daniel cell is `1.1V`. Calculate the standard Gibbs energy of - YouTube

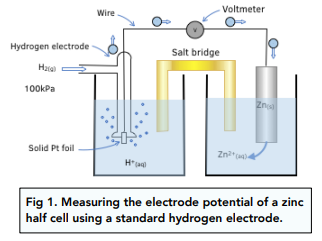
Standard Cell Potential: Calculations, Electron Flow & Feasibility (5.4.2) | CIE A Level Chemistry Revision Notes 2022 | Save My Exams

equilibrium - Calculate the cathode electrode potential in this redox reaction - Chemistry Stack Exchange
![PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/56964684a624c5af38c7e62256db3faa4c542d88/19-Table2-1.png)
PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar

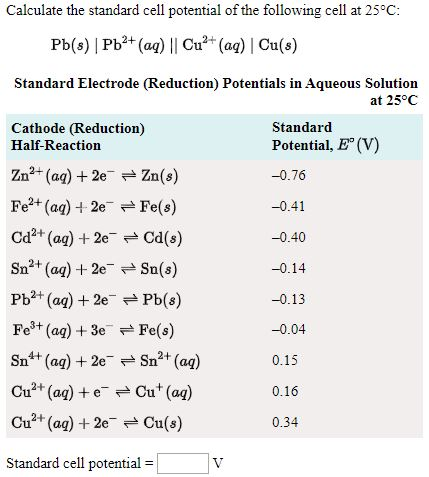
SOLVED: Calculate the standard cell potential of the following cell at 25'C: Pb(s) Pb2+ (aq) || Cu? + (aq) Cu(s) Standard Electrode (Reduction) Potentials in Aqueous Solution at 258C Cathode (Reduction) Half-Reaction