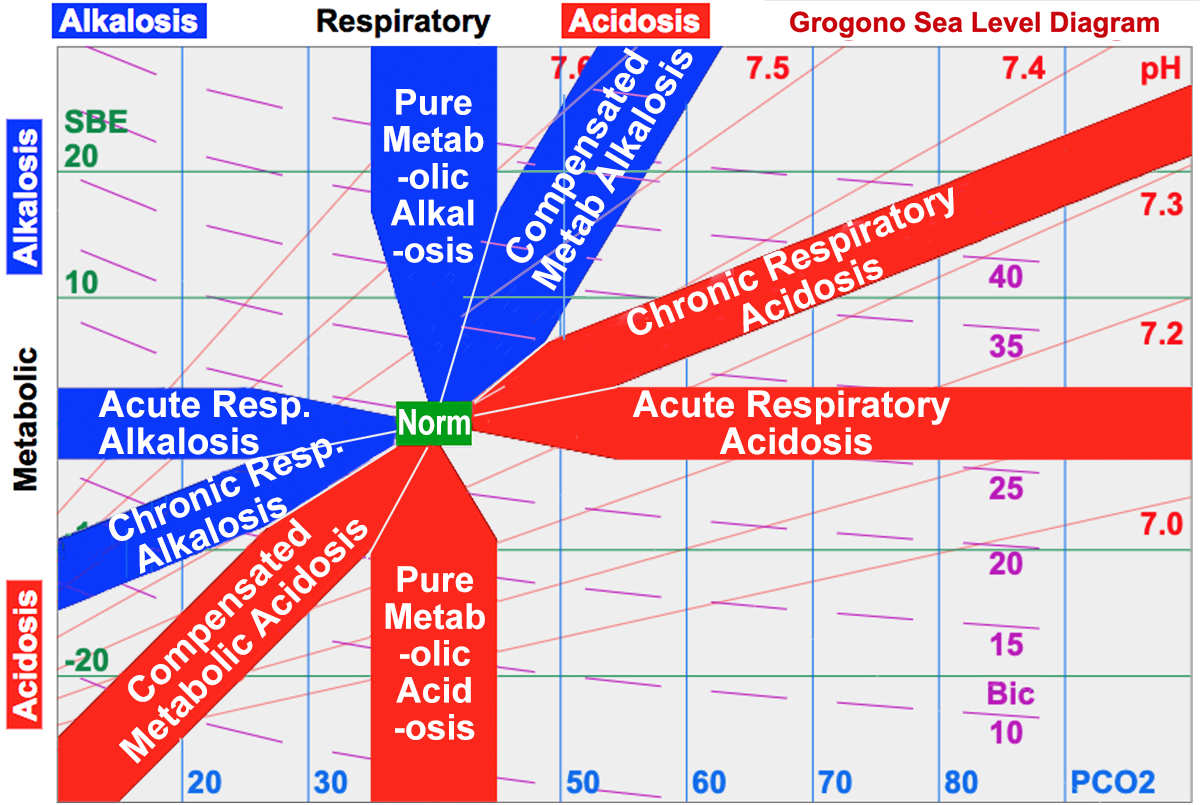
ABG INTERPRETATION. BE = from – 2.5 to mmol/L BE (base excess) is defined as the amount of acid that would be added to blood to titrate it to. - ppt download

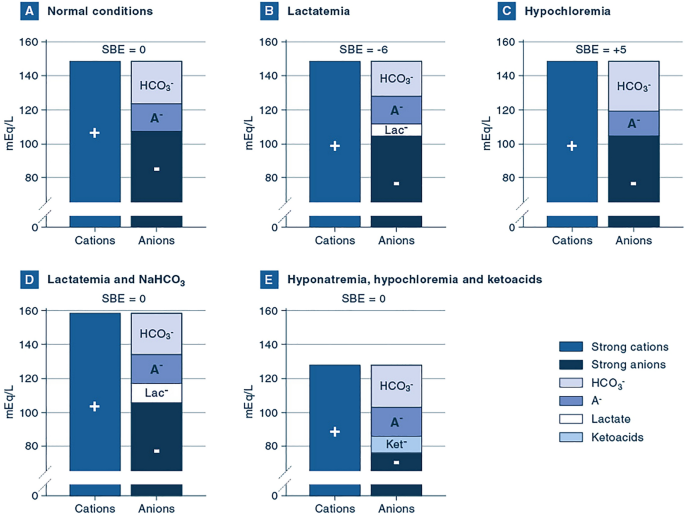
PaCO 2 /pH relationships in vivo. SBE is standard base excess (mmol/L).... | Download Scientific Diagram
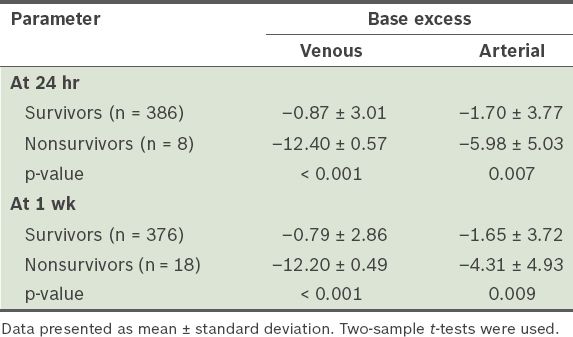
Can venous base excess replace arterial base excess as a marker of early shock and a predictor of survival in trauma? | SMJ

ABG INTERPRETATION. BE = from – 2.5 to mmol/L BE (base excess) is defined as the amount of acid that would be added to blood to titrate it to. - ppt download
![Acid-Base balance Prof. Jan Hanacek. pH and Hydrogen ion concentration pH [H+] nanomol/l ppt download Acid-Base balance Prof. Jan Hanacek. pH and Hydrogen ion concentration pH [H+] nanomol/l ppt download](https://images.slideplayer.com/21/6241563/slides/slide_11.jpg)
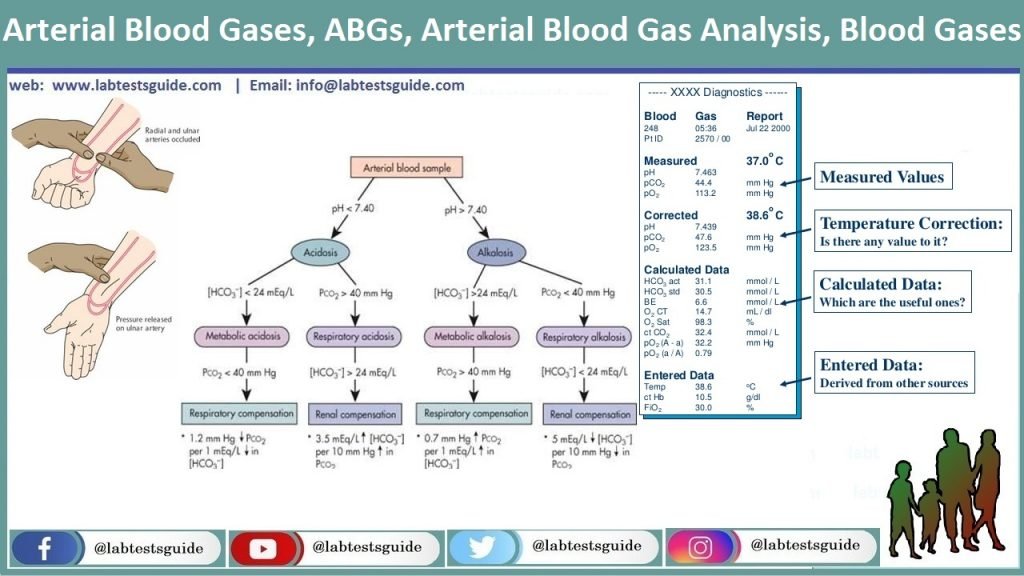
Acid-Base balance Prof. Jan Hanacek. pH and Hydrogen ion concentration pH [H+] nanomol/l ppt download

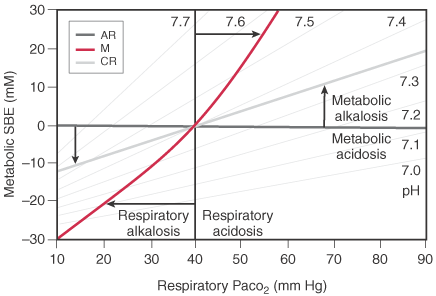

![PDF] Base Excess : the basics October 2017 | Semantic Scholar PDF] Base Excess : the basics October 2017 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f8a9a879c3efdd0170a857700d730c32f8347d2/3-TableI-1.png)
![PDF] Base Excess : the basics October 2017 | Semantic Scholar PDF] Base Excess : the basics October 2017 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f8a9a879c3efdd0170a857700d730c32f8347d2/5-TableIII-1.png)
![PDF] Base Excess : the basics October 2017 | Semantic Scholar PDF] Base Excess : the basics October 2017 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f8a9a879c3efdd0170a857700d730c32f8347d2/6-TableIV-1.png)