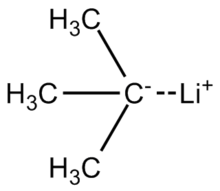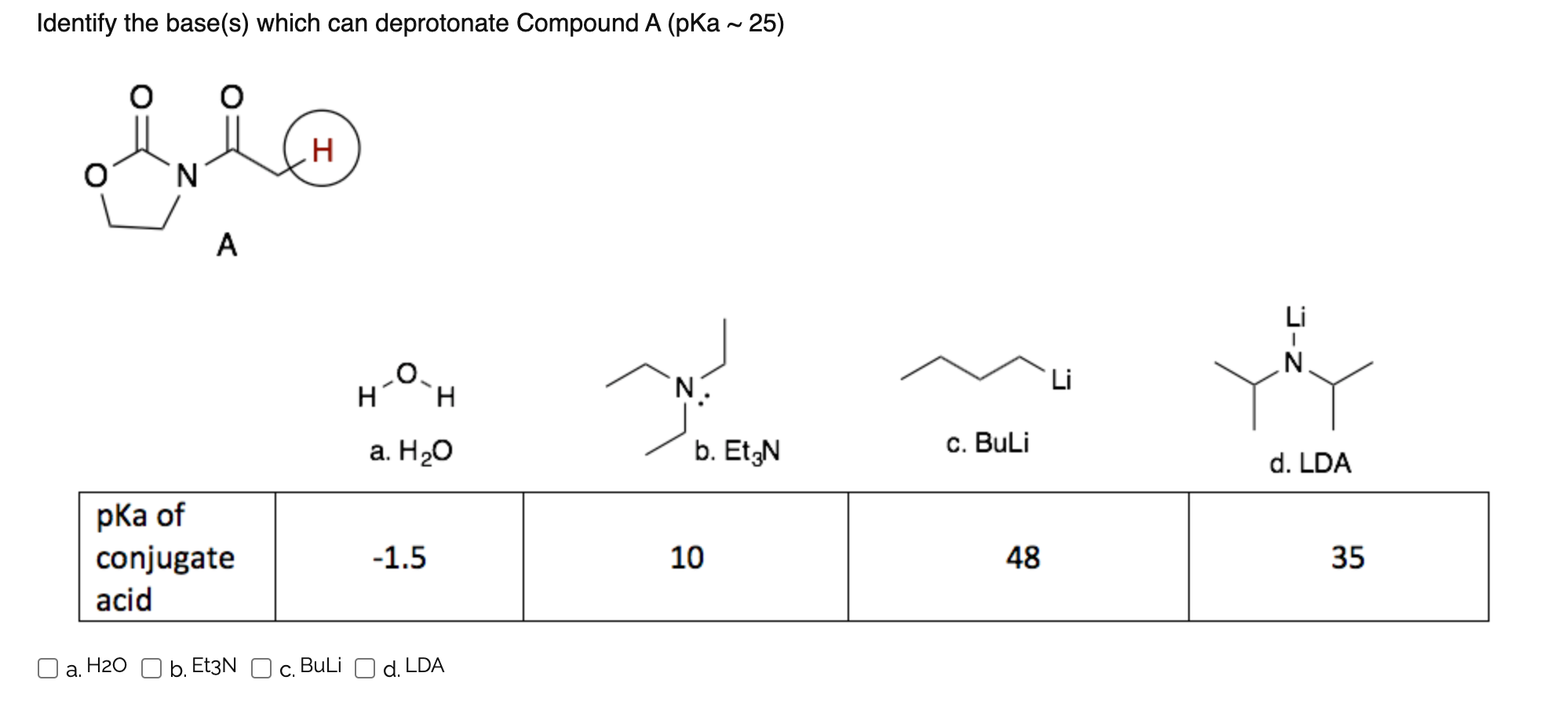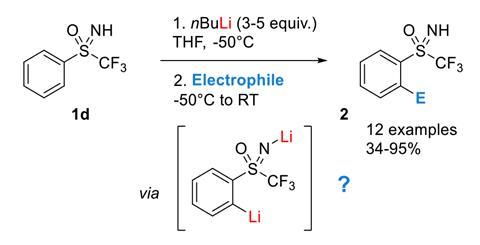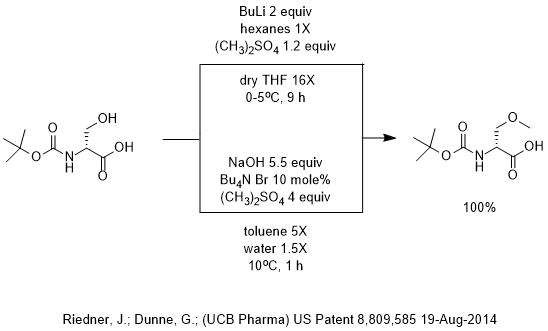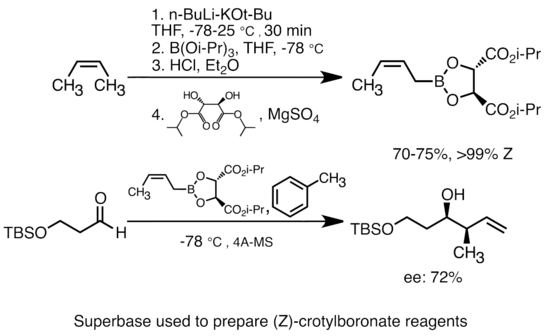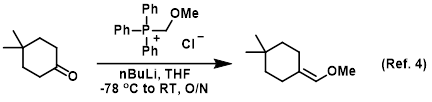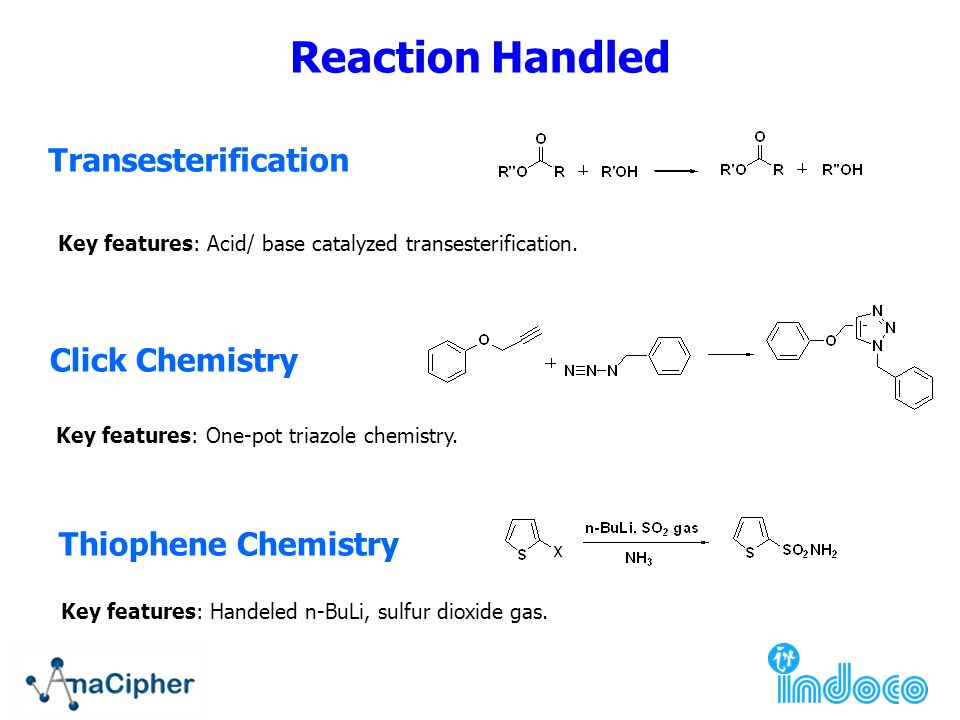
Grignard Reaction Key features: Handling of air/ moisture sensitive chemicals, formation of C-C bond. n-Butyl lithium Key features: Strong base such as. - ppt download
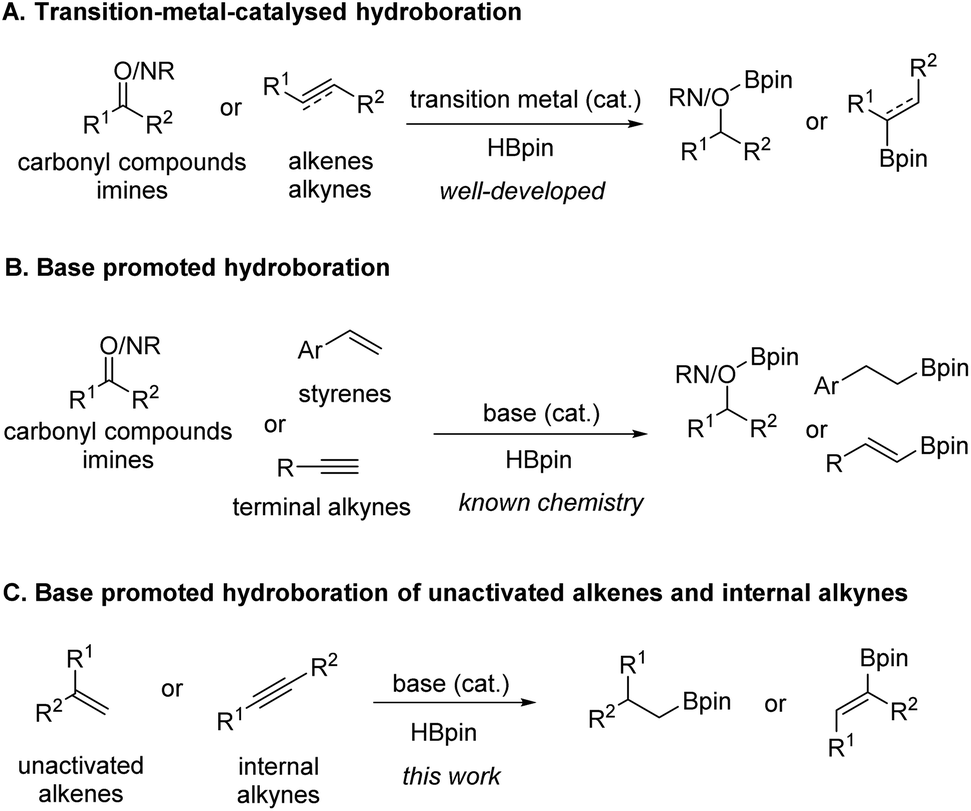
n BuLi-promoted anti -Markovnikov selective hydroboration of unactivated alkenes and internal alkynes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO00750D

Mechanism of the Deprotonation Reaction of Alkyl Benzyl Ethers with n‐Butyllithium - Raposo - 2013 - Chemistry – A European Journal - Wiley Online Library