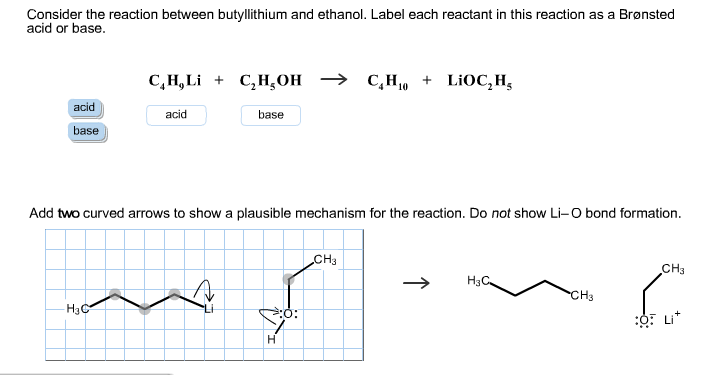
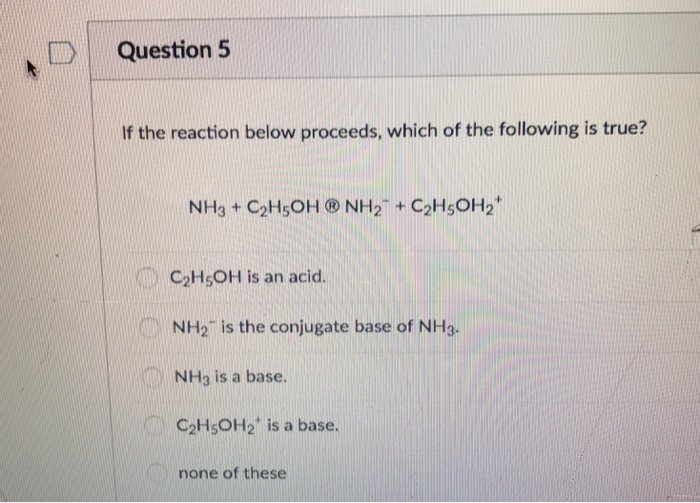
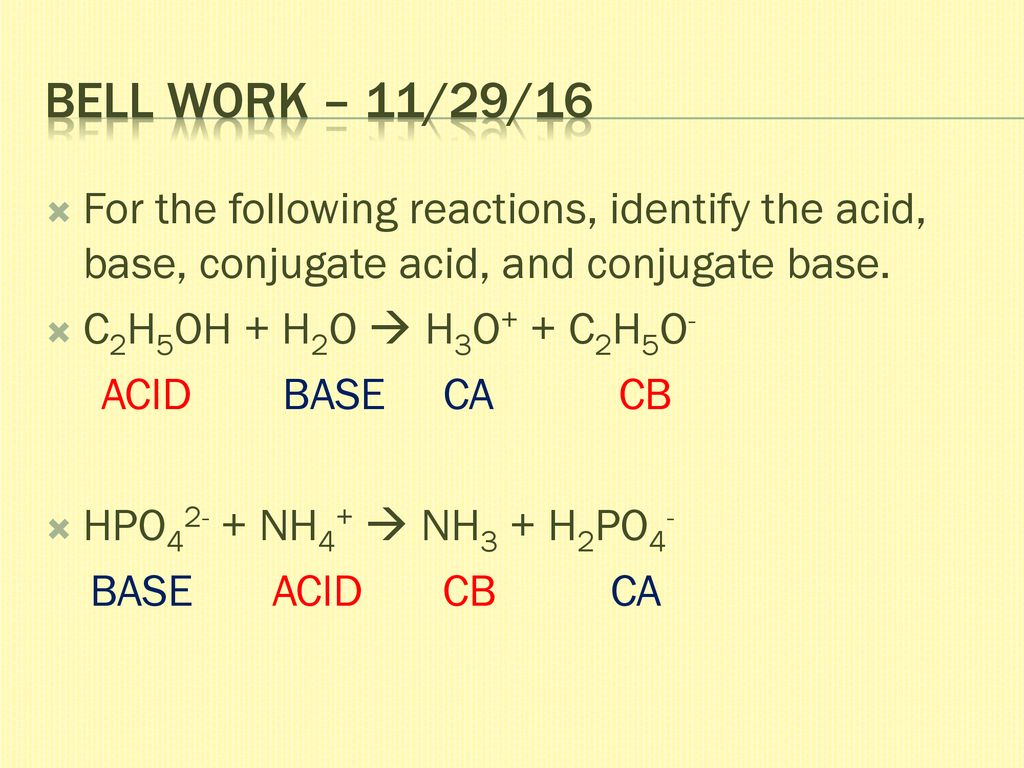
SOLVED: C2H5OH is an substance and a a. organi, non, electrolyte b. organic, strong electrolyte c. strong base, strong electrolyte d. strong acid, strong electrolyte

TOPIC: pH Do Now: Identify as acid, base, salt, or covalent Electrolyte or Not 1. NaCl 6. CaI2 2. C2H5OH 7. HF 3. H2SO4 8. Mg(OH)2 4. NaOH 9. NH3 5. C12H22O ppt download

In an experiment starting with 1 mol C(2)H(5)OH, 1 mol CH(3)COOH, and 1 mol of water, the equilibrium mixture mixture of analysis showa that 54.3% of the acid is eaterified. Calculate K(c).

Reagents and conditions: (a) CCl3CH(OH)2, NH2OH. HCl, 45min heating;... | Download Scientific Diagram

SOLVED: Identify each substance as an Arrhenius acid, an Arrhenius base, or neither. NaOH C2H5OH H3PO4
Which of the following reacts is not shown by formic acid? Reaction with Ca(OH)2 Reaction with I2 / Red P Reaction with NaHCO3 Reaction with C2H5OH

1. Which of the following is not both a Bronsted-Lowry acid and a Bronsted-Lowry base? A) HSO4- (hydrogen sulfate) B) H2PO4- (dihydrogen phosphate) - ppt download