SOLVED: 16- Consider a solution of 2.0 M HCN and 1.0 M NaCN (Ka for HCN = 6.2 10 10). Which of the following statements is true? A) The solution is not

SOLUTION: NaCH3COO, HCl, HCN, NaOH, NH3, NaCN, KNO3, H2SO4, NH4Cl, H2SO3, NaHCO3, Na3PO4 and CH3COOH - Studypool

Exam 3 Practice Problems - Sample Problems (Acid/Base and Solubility) CHM Determine if the following - StuDocu
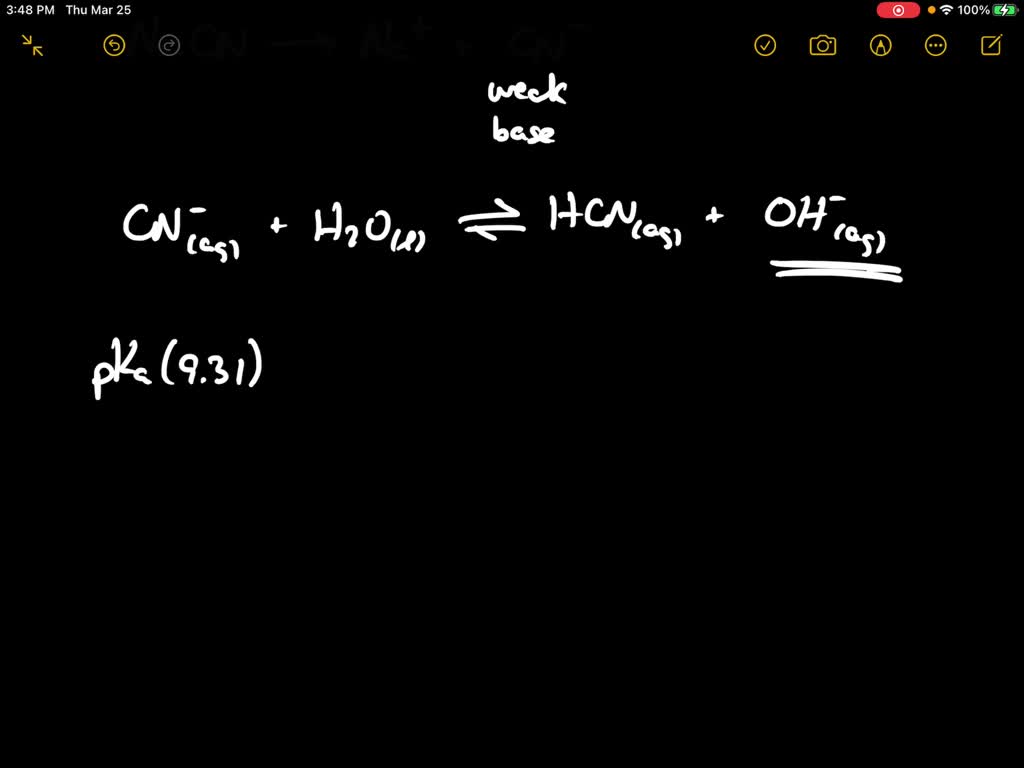
SOLVED:When NaCN dissolves in water, the resulting solution is basic. Account for this observation given that p Ka for HCN is 9.31.
What is the % hydrolysis of NaCN in N/80 solution when dissociation constant for HCN is 1.3 x 10^-9 & Kw = 1 x 10^-14 - Sarthaks eConnect | Largest Online Education Community

Acids Lesson 6 Acid Rain & Hydrolysis. Acid Rain The cause of Acid Rain is the release of acid anhydrides into the environment. Acid Anhydrides are nonmetal. - ppt download